Basic Characteristics
- Electric charge can explain static electricity
- There are only two types of charges, one positive and one negative
- We know this because when certain materials are rubbed together in specific ways, one always produces a positive charge and other a negative.
- Like charges repel, while unlike attract.
- The force between charges decrease with distance
Charge carried by Electrons and Protons
Nearly all charge in nature is carried by protons and electrons, where protons have a positive charge and electrons have a negative charge.
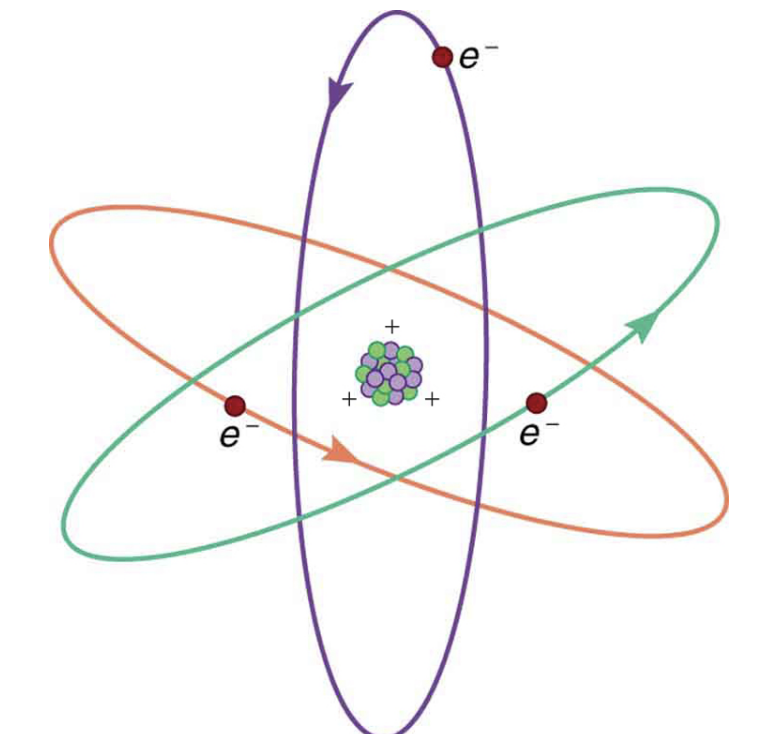 a simplified diagram of an atom, with positively charged protons in the nucleus and negatively charged electrons orbiting. Note: this is a very simplified diagram. real atoms would look absolutely nothing like this.
a simplified diagram of an atom, with positively charged protons in the nucleus and negatively charged electrons orbiting. Note: this is a very simplified diagram. real atoms would look absolutely nothing like this.
The charge of an electron and the charge of a proton are equal in magnitude but opposite in sign (question: is charge a Vector, in that case?). All overall charges are made up of, then, the fundamental basic unit of charge:
Note: the symbol is commonly used for charge and the subscript represents the charge of a single electron and proton. (question: shouldn’t this be negative for electrons? ans: there is an absolute value). The SI Unit for charge is the coulomb, or (C). Therefore, the number of protons needed to make a charge of 1.00 C is:
There is no directly observed charge that is smaller than .
There is no substructure inside of an electron. It is no more divisible. The proton, on the other hand, is composed of three Quarks. Two quarks are known as “up” quarks, and one is known as a “down” quark. The charge of the up quark is , while a down quark is . Therefore, the charge of a proton is:
Law of Conservation of Charge: Total charge is constant in any process.
References
- [OpenStax College Physics 2e][https://assets.openstax.org/oscms-prodcms/media/documents/College_Physics_2e-WEB_7Zesafu.pdf]